What Are PROTAC Linkers?
PROTAC linkers are the connecting units used to assemble proteolysis-targeting chimeras, or PROTACs. A typical PROTAC molecule contains three functional parts: a ligand for the protein of interest, a ligand for an E3 ubiquitin ligase, and a linker that joins the two binding elements into one bifunctional degrader. Although the linker is sometimes viewed as a spacer, it has a direct influence on molecular geometry, ternary complex formation, physicochemical profile, and degradation performance. For teams working in targeted protein degradation, choosing the right PROTAC linker is therefore not a simple synthetic decision; it is a central design variable that can determine whether a degrader is useful for research.
BOC Sciences provides PROTAC linker products and related technical support for research groups developing degraders, building libraries, comparing E3 ligase recruitment strategies, or optimizing linker length and composition. Our product-oriented approach helps medicinal chemistry, chemical biology, and discovery teams move from early concept to practical compound selection with clearer design logic and more efficient procurement planning.
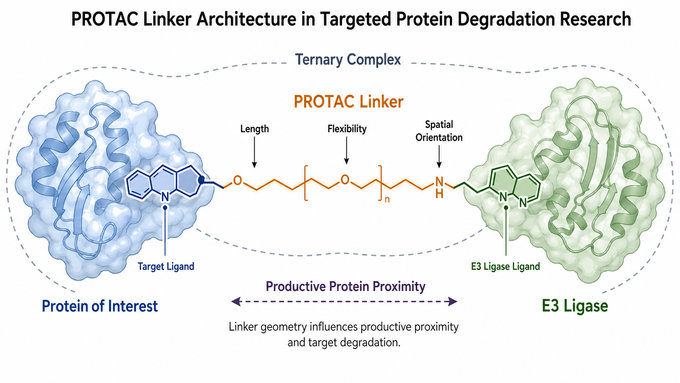 Fig.1 PROTAC Linker Architecture in Targeted Protein Degradation Research (BOC Sciences Original).
Fig.1 PROTAC Linker Architecture in Targeted Protein Degradation Research (BOC Sciences Original).
Definition and Role in PROTAC Architecture
In PROTAC architecture, the linker defines the distance and orientation between two ligands that must act together. The target-binding ligand recognizes the protein of interest, while the E3 ligase ligand recruits an E3 ligase complex. The linker holds these binding elements in a spatial relationship that allows the two proteins to form a productive ternary complex.
- Spatial spacing: The linker determines how far the target ligand and E3 ligase ligand are positioned from each other in the bifunctional degrader.
- Orientation control: The linker influences whether the two recruited proteins can form a productive ternary complex rather than an unproductive encounter.
- Optimization flexibility: The linker can be modified to explore length, polarity, rigidity, and attachment chemistry during degrader development.
- Research relevance: The linker helps connect chemical structure with measurable degradation activity in PROTAC studies.
Linkers also influence properties that are important during compound handling and assay design, including solubility, polarity, molecular size, conformational flexibility, and synthetic accessibility. As a result, linker design is frequently explored through parallel synthesis and structure–activity relationship studies rather than a single fixed design. Common search intentions such as "PROTAC linker design," "PROTAC linker length," and "PROTAC linker library" all reflect the same practical need: researchers require a rational way to compare spacer structures and identify candidates suitable for their degrader series.
Why PROTAC Linker Design Matters?
PROTAC linker design matters because degradation is not driven by binding affinity alone. A degrader must bring the protein of interest and an E3 ligase into a ternary complex in which ubiquitin transfer can occur efficiently. The linker helps organize that complex. It affects whether the two ligands can bind simultaneously, whether the protein surfaces are presented in a favorable orientation, and whether accessible lysine residues on the target are positioned near the ubiquitination machinery.
Linker Length, Flexibility, and Spatial Orientation
Linker length is one of the first parameters explored in PROTAC optimization. Short linkers may create compact degraders but can restrict the ability of the two proteins to approach each other productively. Long linkers may bridge greater distances but can introduce conformational freedom, increase molecular size, and weaken effective complex organization. Flexible linkers such as PEG segments may help a molecule sample multiple conformations, while rigid or semi-rigid linkers may help present the two ligands in a more defined orientation.
Impact on Ternary Complex Formation and Target Degradation
The linker can change ternary complex stability, cooperativity, and selectivity, even when binary binding to the target and E3 ligase remains measurable. In successful PROTAC design, the linker supports a complex that allows ubiquitin transfer and subsequent proteasomal degradation of the target protein. In less productive designs, the molecule may still bind both proteins but fail to position them effectively. This explains why linker screening is often required after identifying suitable ligand pairs.
For projects that require deeper design input, BOC Sciences offers PROTAC design services to help researchers evaluate ligand pairing, linker selection, conjugation strategy, and degrader optimization routes. This support is especially valuable when a team has promising ligands but needs to translate them into a practical degrader series.
Structural Features of PROTAC Linkers
The structural features of a PROTAC linker determine more than physical distance. They affect how the degrader behaves in solution, how it samples conformations, how it interacts with protein surfaces, and how easily it can be synthesized and modified. An effective linker strategy considers both the chemical structure of the linker itself and the way it is attached to each ligand.
Chemical Composition and Functional Handles
PROTAC linkers may contain PEG repeats, aliphatic chains, alkyl-ether segments, amides, carbamates, triazoles, piperazines, heteroaryl rings, aromatic rings, or cleavable motifs. The selection depends on desired polarity, flexibility, stability in the assay context, and compatibility with the available ligand handles. Functional handles are particularly important for synthesis because they define how the linker will be attached to the target ligand and E3 ligase ligand. When the ligand pair is already defined, handle compatibility can become the first selection filter before length, rigidity, or polarity are optimized.
Common structural and functional elements include:
- PEG Repeats
- Alkyl Chains
- Alkyl-Ether Segments
- Amide Groups
- Triazole Units
- Piperazine Units
- Heteroaryl Rings
- Aromatic Rings
Rigidity, Polarity, and Conformational Control
Rigidity and polarity shape the conformational behavior of PROTAC molecules. PEG linkers are generally flexible and hydrophilic, supporting solubility and distance scanning. Alkyl chains add hydrophobic character and can sometimes support cell-based research where excessive polarity is undesirable. Aromatic, heteroaryl, piperazine, and triazole-containing linkers can introduce conformational control and defined orientation, which may improve ternary complex organization for certain ligand pairs. The goal is not to maximize one property, but to balance molecular shape and handling characteristics for the intended research workflow.
Key design variables usually include:
- Flexible Spacing
- Defined Geometry
- Balanced Polarity
- Conformational Control
Common Types of PROTAC Linkers
PROTAC linker chemistry is diverse, but several linker families appear frequently in degrader design workflows. Each linker type offers different benefits and limitations. Researchers often compare more than one class because the optimal linker cannot always be predicted from ligand structures alone.
| Linker Type | Key Structural Character | Typical Research Use | Selection Notes |
| PEG Linkers | Flexible, oxygen-rich, hydrophilic chains | Length scanning, solubility adjustment, early degrader exploration | Useful when flexibility and aqueous compatibility are needed |
| Alkyl Linkers | Hydrophobic carbon chains with variable length | Hydrophobic balance, compact degrader design, permeability-oriented studies | May require careful balancing of solubility and aggregation risk |
| Alkyl-Ether Linkers | Mixed hydrophobic and polar spacing units | Intermediate profiles between PEG and alkyl chains | Helpful when neither fully PEG nor fully alkyl linkers fit the project |
| Rigid and Semi-Rigid Linkers | Aromatic, heteroaryl, cyclic, triazole, or piperazine elements | Defined geometry, conformational restriction, ternary complex tuning | Often explored when flexible linkers give weak or inconsistent degradation |
| Cleavable Linkers | Motifs designed to respond to selected chemical or biochemical conditions | Conditional release concepts and mechanistic research tools | Require careful matching to the intended assay environment |
PEG Linkers for Flexible and Hydrophilic Spacing
PEG linkers are widely used in PROTAC research because they offer flexible spacing and increased hydrophilicity. They are useful for early linker-length evaluation, especially when the preferred distance between the two ligands is unknown. By comparing different PEG repeat numbers, researchers can explore how the degrader responds to changes in reach, polarity, and conformational mobility.
They are commonly selected for:
- Flexible Spacing
- Hydrophilic Character
- Length Scanning
- Library Construction
Alkyl and Alkyl-Ether Linkers for Hydrophobic Balance
Alkyl linkers provide hydrophobic spacing and can reduce the polar surface contribution introduced by PEG units. Alkyl-ether linkers provide an intermediate option by combining carbon-chain spacing with oxygen atoms that tune polarity. These linkers can be valuable when a degrader needs a more balanced physicochemical profile.
Typical design considerations include:
- Hydrophobic Balance
- Compact Spacing
- Reduced Polarity
- Mixed Ether Design
Cleavable Linkers for Conditional Release Strategies
Cleavable linkers contain motifs that can be disconnected under defined chemical or biochemical conditions. In PROTAC research, they may be used to investigate release mechanisms, conditional degrader behavior, or tool-compound concepts. Cleavable linker design requires careful consideration because the linker must remain stable during synthesis, storage, and the relevant assay period while still responding to the intended trigger.
These projects often require attention to:
- Assay Compatibility
- Handling Stability
- Trigger Selection
- Mechanistic Research
Rigid and Semi-Rigid Linkers for Defined Geometry
Rigid and semi-rigid linkers use aromatic rings, heteroaryl groups, piperazine, triazole, or cyclic structures to control shape. These linkers can reduce excessive conformational freedom and help present ligand motifs in a more defined orientation. They are often explored after flexible linkers fail to produce strong degradation or when structural modeling suggests that a specific angle may favor target–E3 alignment.
Representative rigid or semi-rigid options include:
- Aromatic Linkers
- Heteroaryl Linkers
- Piperazine Linkers
- Triazole Linkers
- Cyclic Linkers
- Semi-Rigid Spacers
Need Reliable PROTAC Linkers for Your Research?
BOC Sciences offers a broad portfolio of in-stock PROTAC linkers to support the design and development of targeted protein degraders. If you cannot find the linker you need or require a customized structure, our team can provide tailored synthesis solutions.
Request a Quote
PROTAC Linker Length and Optimization Strategy
PROTAC linker length is one of the most frequently searched and practically important topics in degrader design. Length is not simply a numeric value; it interacts with linker flexibility, exit vector direction, ligand size, protein surface topology, and assay conditions. A rational linker optimization strategy usually begins with a focused set of lengths and chemotypes, followed by iterative refinement based on degradation readouts, binding data, and physicochemical observations.
Short, Medium, and Long Linker Considerations
Short linkers may produce compact degraders and reduce molecular size, but they can also prevent simultaneous binding if the target and E3 ligase surfaces require more separation. Medium linkers often provide a practical starting region because they allow distance exploration without excessive size. Long linkers may be useful when the ligand exit vectors are far apart in the productive ternary complex, but additional length can increase flexibility and reduce the probability of adopting the correct conformation. The most informative strategy is often to test a small range of short, medium, and longer linkers with matched attachment chemistry.
Attachment Position and Exit Vector Selection
The same linker can produce different behavior when attached through different positions on a ligand. Exit vector selection should preserve essential binding interactions while orienting the linker toward solvent-exposed space or a protein-protein interface that supports ternary complex formation. For E3 ligase binders, exit vectors are often chosen from chemically established positions; for target ligands, they may require careful medicinal chemistry evaluation. BOC Sciences can support ligand design for E3 ligase projects and ligand design for target protein workflows when teams need to evaluate modification points before linker attachment.
Balancing Permeability, Solubility, and Molecular Size
PROTAC molecules are typically larger and more complex than conventional small-molecule probes. Linker optimization must therefore consider molecular size, polar surface area, hydrogen-bonding capacity, rotatable bonds, and solubility. PEG linkers may help solubility but add polarity and flexibility. Alkyl linkers may reduce polarity but increase hydrophobic character. Rigid linkers may improve shape control but can affect synthetic route and compound handling. The goal is not to maximize one property; it is to balance properties in a way that supports the specific research assay and degrader objective.
When to Explore Minimal-Linker or Linker-Free Designs?
Minimal-linker or linker-free PROTAC concepts may be considered when two ligand motifs can be joined directly or through a very short connector without losing productive orientation. These designs can reduce molecular size and may simplify structure, but they are not automatically superior. They require suitable ligand exit vectors and a geometry that still allows target and E3 ligase engagement. For many projects, minimal-linker designs are evaluated alongside classical PEG, alkyl, and rigid linker options to determine whether simplified architecture supports the desired degradation profile.
Mechanistic Role of Linkers in Targeted Protein Degradation
The mechanistic role of a PROTAC linker becomes clear when the degradation cycle is viewed as a sequence of coordinated events. The degrader first engages one or both binding partners, then forms a ternary complex, supports ubiquitin transfer to the target protein, and allows the target to be directed to the proteasome. The linker contributes to this process by influencing geometry, residence of the ternary complex, and productive positioning of protein surfaces.
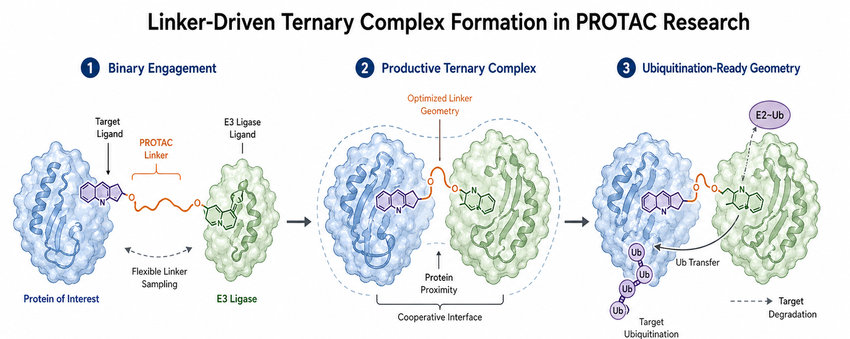 Fig.2 Linker-Driven Ternary Complex Formation in PROTAC Research (BOC Sciences Original).
Fig.2 Linker-Driven Ternary Complex Formation in PROTAC Research (BOC Sciences Original).
Bringing the Protein of Interest and E3 Ligase into Productive Proximity
The first mechanistic requirement is productive proximity. The linker must connect the target ligand and E3 ligase ligand in a way that allows both proteins to occupy the same assembly without severe steric conflict. Productive proximity is not only about distance; it also depends on the angle and orientation in which each protein is presented.
Supporting Ubiquitination Efficiency Through Proper Complex Geometry
After ternary complex formation, the target protein must be positioned so that accessible lysine residues can receive ubiquitin chains. Linker geometry can influence whether the target is oriented favorably relative to the E2 enzyme and E3 ligase complex. Complementary services such as protein ubiquitination services and degradation ability assay support can help connect chemical design with functional observations.
Influencing Cooperativity, Selectivity, and Degradation Profiles
A productive PROTAC may show positive cooperativity when the target and E3 ligase stabilize each other within the ternary complex. Linker composition can influence this cooperativity by enabling favorable protein-protein contacts or reducing unfavorable orientations. Measuring binding behavior through binding affinity measurement can provide useful context for interpreting degrader performance.
Avoiding Unproductive Binary Binding and Hook-Related Effects
At certain concentrations, a degrader may favor binary binding to one partner rather than productive ternary complex formation. This can reduce degradation efficiency and may contribute to hook-related behavior in some systems. Linker optimization helps reduce these unproductive states by promoting a geometry that favors ternary assembly over isolated binary interactions.
Applications of PROTAC Linkers in Research
PROTAC linkers are not only building blocks for individual degraders. They are enabling components for screening collections, SAR campaigns, chemical biology tools, and platform development. By varying linker length and chemistry, researchers can learn how target–E3 pairing responds to spatial changes and can identify degrader designs that merit deeper study.
PROTAC Discovery and Structure–Activity Relationship Studies
In PROTAC discovery, linkers are often varied systematically to generate a structure–activity relationship across length, polarity, rigidity, and attachment position. This approach helps separate ligand-binding limitations from linker-driven ternary complex limitations. A degrader series may show weak activity with one linker type and stronger activity with another, even when the same target and E3 ligase ligands are used. Linker SAR therefore provides actionable design information early in a degrader program.
Degrader Library Construction and Screening Workflows
Linker libraries help teams build diverse degrader collections more efficiently. A well-planned linker library can include PEG, alkyl, alkyl-ether, rigid, semi-rigid, and functionalized variants that support multiple conjugation routes. These libraries are useful for parallel synthesis, hit follow-up, and early screening workflows where researchers need to explore chemical space without designing every linker from the beginning.
E3 Ligase Recruitment and Target Protein Validation
Different E3 ligase ligands may require different linker positions and spacer properties. A linker that works well with one E3 system may not suit another because the ligase surface, ligand exit vector, and ternary complex topology can change. PROTAC linkers support comparative studies across E3 ligase recruitment strategies and help researchers assess whether a target protein can be degraded through a selected ligase pathway.
Chemical Biology Tools for Protein Degradation Research
PROTAC linkers are used in chemical biology to develop tool degraders, negative controls, photoresponsive concepts, covalent degrader formats, and mechanistic probes. Functionalized linkers can also support labeling, enrichment, or modular analog preparation when compatible with the study design. These research tools help teams investigate protein function, degradation kinetics, and degrader mechanism in a controlled experimental setting.
How to Select PROTAC Linkers for Your Project?
Selecting a PROTAC linker requires a balance between chemical feasibility and biological function. The starting point should be the ligand pair, the known or predicted attachment points, and the intended assay format. From there, researchers can choose a focused set of linkers that explore meaningful variations in length, polarity, rigidity, and functional handle compatibility.
| Selection Factor | Key Question | Practical Linker Direction |
| Ligand Pair | Do both ligands tolerate modification at the selected positions? | Confirm exit vectors before expanding linker length |
| Distance Requirement | How far apart should the two binding motifs be presented? | Compare short, medium, and longer linkers in a matched series |
| Flexibility | Does the complex need conformational sampling or defined geometry? | Use PEG for flexible scanning; use rigid elements for orientation control |
| Polarity | Is the molecule too hydrophobic or too polar for the assay system? | Adjust PEG, alkyl, ether, amide, or heteroatom content |
| Conjugation Chemistry | Which functional handles are compatible with the synthesis route? | Select amine, acid, azide, alkyne, alcohol, or activated handles accordingly |
| Optimization Stage | Is the project in discovery, hit follow-up, or focused refinement? | Use broader linker diversity early and more focused analogs later |
Selecting Linkers Based on Ligand Pair and Binding Geometry
Begin by examining the ligand pair. The target ligand and E3 ligase ligand should each have a modification site that is compatible with binding. When structural information is available, solvent exposure and exit vector direction can guide initial linker selection. When structural information is limited, a broader linker scan may be needed. The most useful first set usually includes linkers that differ in length and flexibility while keeping the attachment chemistry consistent.
Matching Linker Chemistry to Assay and Solubility Needs
Assay context should guide linker chemistry. Biochemical assays may tolerate linkers that behave differently from those used in cellular research. PEG linkers can improve aqueous compatibility, while alkyl linkers can reduce excessive polarity. Heteroatom-containing or cyclic linkers can tune shape and solubility together. The selected linker should support compound handling, concentration preparation, and reliable interpretation of degradation readouts.
Comparing PEG, Alkyl, and Rigid Linker Options
A practical comparison set may include one or more PEG linkers, alkyl or alkyl-ether linkers, and a rigid or semi-rigid linker. This allows researchers to evaluate whether the degrader benefits from flexibility, hydrophobic balance, or defined geometry. The comparison is most informative when other variables are minimized. For example, keeping both ligands and attachment positions constant while changing only the linker helps reveal the true impact of linker composition.
Planning Iterative Linker Screening and Optimization
Linker optimization should be iterative. Early screening identifies workable length and chemotype ranges. Follow-up designs refine polarity, rigidity, and attachment chemistry. Later analogs may focus on reducing molecular size, improving selectivity, or simplifying synthesis. BOC Sciences can support teams that need catalog linker selection, custom linker preparation, or a coordinated plan for building PROTAC analogs around a defined research objective.
PROTAC Linker Library and Product Options
BOC Sciences offers PROTAC linker products designed to support degrader synthesis, library construction, conjugation chemistry, and linker optimization. Product options may include flexible PEG linkers, hydrophobic alkyl linkers, alkyl-ether linkers, rigid aromatic linkers, heterocyclic linkers, and linkers bearing terminal functional groups for modular coupling. These products help researchers reduce early design uncertainty and build degrader series with clearer structural variation.
Ready-to-Use Linkers for Degrader Synthesis
Ready-to-use linkers are suitable for teams that already know the ligand pair and need practical building blocks for conjugation. These linkers can support early screening, analog expansion, and focused SAR. Researchers can select by length, chemotype, terminal handle, and intended conjugation route. BOC Sciences helps customers evaluate linker options that fit the target ligand, E3 ligase ligand, and desired degrader design.
Linkers with Reactive Groups for Conjugation Chemistry
Functionalized linkers contain reactive groups that simplify assembly with ligand motifs. Terminal amines, acids, azides, alkynes, alcohols, and activated groups can be matched with complementary handles on target ligands or E3 ligase ligands. These options allow researchers to choose chemistry that aligns with available intermediates and synthetic constraints while still exploring length and composition.
Linkers for E3 Ligase Ligand–Linker Conjugates
E3 ligase ligand–linker conjugates can streamline degrader synthesis by providing one half of the PROTAC architecture in a prepared form. This approach is useful when researchers want to combine a selected E3 ligase recruiting motif with multiple target ligands. Linker selection remains important because the conjugate must present the E3 ligand in a way that allows productive recruitment and compatible target-ligand coupling.
Linkers for Target Ligand–Linker Conjugates
Target ligand–linker conjugates support the opposite modular strategy: a target-binding motif is connected to a linker and then coupled with one or more E3 ligase ligands. This can be useful when the target ligand modification point is challenging or when a research team wants to compare several E3 ligase recruitment options while keeping the target side constant. BOC Sciences can support both catalog selection and customized linker planning for these workflows.
Why Choose BOC Sciences PROTAC Linker Products?
BOC Sciences supports PROTAC linker research with a product portfolio and service capabilities aligned with real discovery workflows. Our focus is to help research teams select linkers that are chemically appropriate, technically relevant, and practical for degrader construction. We avoid one-size-fits-all recommendations because linker performance depends on the ligand pair, protein geometry, assay context, and optimization stage.
 Broad Linker Selection for Diverse Degrader Designs
Broad Linker Selection for Diverse Degrader Designs
BOC Sciences provides a wide selection of PROTAC linkers covering PEG, alkyl, alkyl-ether, rigid, semi-rigid, heteroatom-containing, and functionalized structures. This diversity allows researchers to compare length, flexibility, polarity, and conjugation handles within a coherent degrader design plan.
 Technical Support for Linker Selection and Project Planning
Technical Support for Linker Selection and Project Planning
Our team can discuss linker selection based on ligand pair, exit vectors, intended assay, solubility needs, and synthesis route. This helps researchers narrow the search space and design more informative analog sets rather than choosing linkers only by availability or chain length.
 Custom Synthesis Support for Specialized Linker Requirements
Custom Synthesis Support for Specialized Linker Requirements
Some degrader programs require linker structures that are not available as catalog products. BOC Sciences can support custom linker and conjugate preparation for projects requiring specialized functional handles, unusual spacing elements, or matched analog series for SAR studies.
 Research-Oriented Product Information for Efficient Procurement
Research-Oriented Product Information for Efficient Procurement
Procurement teams and project scientists need clear product information to make confident ordering decisions. BOC Sciences emphasizes practical details such as linker class, reactive handle, chemical compatibility, and potential research use so that product selection aligns with experimental planning.
Frequently Asked Questions (FAQ)

Still have questions?
Contact Us
How do I choose the right PROTAC linker?
Choosing the right PROTAC linker depends on the target protein, E3 ligase ligand, warhead, desired ternary complex geometry, and overall physicochemical profile of the degrader. Researchers often compare linker length, rigidity, polarity, solubility, and attachment chemistry to understand how each factor influences degradation potency and selectivity. For early projects, it is common to evaluate a focused panel of alkyl, PEG, or semi-rigid linkers before moving into more refined structure-activity studies.
What linker properties affect targeted protein degradation?
Linker length, flexibility, hydrophilicity, steric profile, and functional handles can strongly influence how a PROTAC molecule brings the target protein and E3 ligase into productive proximity. A linker that is too short, too long, too rigid, or too flexible may reduce ternary complex formation or cellular activity. Scientists also consider permeability, solubility, and metabolic stability when comparing linker designs. BOC Sciences supports drug discovery researchers with diverse PROTAC linker building blocks for these exploratory studies.
Which PROTAC linkers suit early discovery projects?
Early discovery projects usually benefit from a chemically diverse linker set rather than a single “best” linker. Researchers may begin with alkyl linkers for hydrophobic spacing, PEG linkers for increased polarity, and functionalized linkers for convenient conjugation. This approach helps reveal whether degradation activity is sensitive to distance, flexibility, or linker composition. A practical screening library can reduce design uncertainty and guide the next round of degrader optimization.
Can PROTAC linkers be customized for my target?
Yes, PROTAC linkers can often be customized based on the target ligand, E3 ligase binder, conjugation site, and desired molecular properties. Customization may involve changing chain length, adding heteroatoms, introducing branching, modifying terminal functional groups, or designing linkers with different rigidity. For complex research needs, BOC Sciences can provide support around linker selection and custom synthesis discussions, helping researchers match chemical design choices with project-specific degradation strategies.
What should buyers consider before ordering PROTAC linkers?
Procurement teams and researchers should consider the intended application, compatibility with planned coupling chemistry, documentation needs, available structural diversity, and whether technical consultation is helpful before purchasing PROTAC linkers. It is also useful to align ordering decisions with the broader degrader design plan, including target ligand availability and E3 ligase strategy. BOC Sciences offers drug development products and technical communication that can help buyers evaluate linker options without overcomplicating early-stage research planning.
Client Feedback on PROTAC Linker Products
Useful Linker Diversity for Early SAR
“The PROTAC linker options from BOC Sciences helped our discovery chemistry team compare PEG, alkyl, and more rigid spacers without rebuilding the sourcing plan for every analog. The product information made it easier to organize a focused linker matrix for our degrader series.”
— Discovery Chemistry Lead, North America
Clear Communication Around Functional Handles
“We needed linkers with compatible reactive groups for a ligand pair that had limited modification positions. BOC Sciences provided helpful technical discussion on handle selection and linker class, which supported a more efficient synthesis planning process.”
— Senior Research Scientist, Europe
Support for Custom Linker Needs
“Our project required a set of related linkers to test length and rigidity effects. The ability to discuss customized structures with BOC Sciences was valuable because our team could align compound design with the questions we wanted to answer experimentally.”
— Project Manager, Research Organization
Helpful for Procurement and Project Coordination
“The linker information was organized in a way that supported both scientific review and purchasing decisions. Our team could compare product classes, understand the intended chemistry, and move the request forward with fewer internal clarification cycles.”
— Procurement Specialist, Life Science R&D
Discover More Research Products
Explore featured products that can expand your research options and accelerate your next discovery.
Expert Services to Move Your Project Forward
Access end-to-end service solutions that help bring efficiency, flexibility, and expertise to your research pipeline.
Insights and Resources
News
Technical Information





















 Fig.1 PROTAC Linker Architecture in Targeted Protein Degradation Research (BOC Sciences Original).
Fig.1 PROTAC Linker Architecture in Targeted Protein Degradation Research (BOC Sciences Original). Fig.2 Linker-Driven Ternary Complex Formation in PROTAC Research (BOC Sciences Original).
Fig.2 Linker-Driven Ternary Complex Formation in PROTAC Research (BOC Sciences Original).

