As a leading service provider in drug discovery and development, BOC Sciences is fully qualified and committed to providing one-stop PROTAC® development services, which has become a promising strategy in the field of small molecular drug discovery. With a comprehensive and advanced platform, we design PROTAC diastereomer to customers around the world to meet new drug discovery goals.
Introduction
Diastereomers are stereoisomers in which molecules have two or more chiral centers and the relationship between molecules is not mirrored. In PROTAC drug development, in order to evaluate the pharmacokinetics of a single diastereomer or mixture of diastereomers, manufacturers should quantitatively analyze samples from the early stages of drug development for a single diastereomer in vivo. This will allow the assessment of the possibility of mutual transformation as well as the distribution of absorption, distribution, biotransformation and excretion (ADBE) of individual isomers. When the drug is a raceme and the pharmacokinetic characteristics of the isomers are different, the manufacturer should monitor the diastereomer separately to determine properties such as dose linearity and the effects of metabolic or excretory changes and drug-drug interactions.
Application
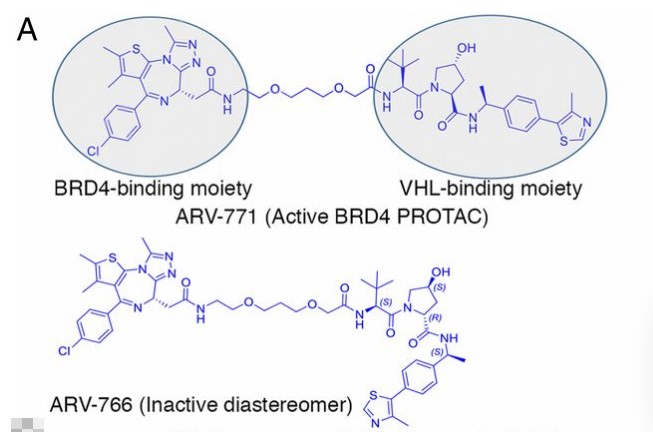
Related studies have proved that ARV-771, is a small molecular pan-BET degrader based on PROTAC technology. This study is the first to prove the efficacy of small molecular BET degradants in solid tumors and may represent an important therapeutic progress in CRPC therapy. In the study, ARV-766 (the diastereomeric isomer of ARV-771, which has the opposite configuration on hydroxyproline and has no affinity for VHL) was designed as the negative control of BET degradation. It is reported that the binding affinity of ARV-771 and diastereomeric ARV-766for BET bromodomains to be comparable to that of Kd of JQ-1 as reported in the literature. However, ARV-766 only showed a small inhibition of c-MYC, which indicated that BET PROTAC had lower cell permeability than JQ-1, and the significant efficacy of ARV-771 was probably caused by the "catalytic" nature of its cellular activity. It shows that the design of diastereomer as negative control is necessary in the study of PROTAC.
Our Advantages
- Diastereomer Design and Optimization
- Creativity and innovation of passionate chemistry researchers
- Process Research, Development, and Optimization
- Advanced Synthetic and Analytical Platform
- Data analysis, detailed report with results and discussion
- Quality one-stop service
References:
- Cromm, P. M., Samarasinghe, K. T., Hines, J., & Crews, C. M. (2018). Addressing kinase-independent functions of Fak via Protac-mediated degradation. Journal of the American Chemical Society, 140(49), 17019-17026.
- Qian, Y., Dong, H., Wang, J., Berlin, M., Crew, A. P., & Crews, C. M. (2017). U.S. Patent Application No. 15/230,354.
* PROTAC® is a registered trademark of Arvinas Operations, Inc., and is used under license.


