Project Background
A biotech client had developed a BRD4-directed degrader with strong cellular DC50 performance and rapid transcriptional suppression in tumor cell lines, but the team lacked clarity on whether this molecule could maintain sufficient exposure and degradation in animals. They needed an in vivo study capable of distinguishing between temporary pathway inhibition and durable target removal in a xenograft setting.
Technical Challenges
The compound showed promising in vitro potency but variable formulation behavior and uncertain tissue retention. The client's key concern was whether intermittent dosing could still sustain deep BRD4 loss inside tumors.
BOC Sciences Solutions
- Study Design Optimization: We established a multi-time-point design covering plasma exposure, tumor collection, target degradation, and pathway biomarkers after single and repeated dosing.
- Formulation and Dosing Comparison: Two formulation approaches and three dosing schedules were screened to identify conditions with the most stable exposure profile.
- Mechanistic Readout Integration: Tumor samples were assessed for BRD4 reduction and downstream transcriptional markers to differentiate transient inhibition from sustained degrader activity.
Project Outcomes
Across 18 evaluated study conditions, one formulation-schedule combination produced the strongest tumor exposure consistency and achieved approximately 75% BRD4 degradation at the optimal sampling window, with durable pathway suppression and clear tumor growth delay relative to comparator settings. The client used these data to prioritize a lead regimen and refine the next-stage efficacy package.
Project Background
A pharmaceutical team was advancing three kinase-targeting PROTAC candidates built on related warheads but different linker architectures and E3 ligase engagement profiles. Their cellular data were encouraging, yet the molecules behaved differently in microsomal stability and permeability experiments. The client needed an in vivo ranking strategy to determine which degrader offered the best balance of exposure, tissue delivery, and functional response.
Technical Challenges
The main challenge was that all three candidates produced measurable cellular degradation, but it was unclear whether the differences would remain meaningful in animals. The client also wanted to know whether weak efficacy for some analogs was caused by insufficient exposure or poor tissue-level degradation efficiency.
BOC Sciences Solutions
- Comparative PK-Efficacy Framework: We designed a side-by-side evaluation package that compared route-dependent exposure, tissue distribution, degradation depth, and efficacy endpoints across all three molecules.
- Target-Class-Informed Interpretation: Prior mechanistic understanding from PROTACs targeting protein kinases was incorporated into study planning to guide biomarker selection and tissue timing.
- Decision-Focused Reporting: Results were organized into a ranking matrix covering exposure durability, degradation onset, recovery pattern, and pharmacological response.
Project Outcomes
The study showed that one candidate consistently outperformed the others, delivering deeper target loss, broader tumor coverage, and the most stable efficacy trend under matched dosing conditions. In total, 3 lead candidates, 2 administration routes, and 4 key biomarker windows were evaluated, enabling the client to eliminate lower-value analogs and concentrate resources on the top-performing degrader architecture.

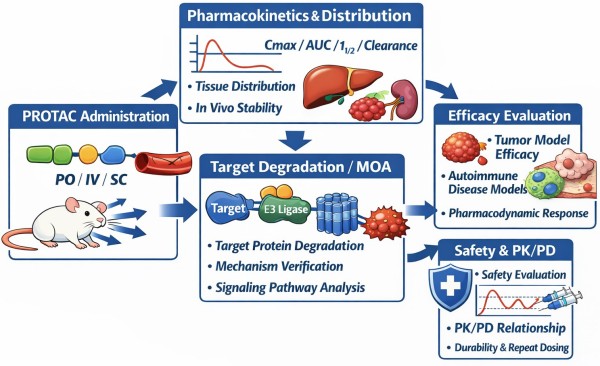 Fig.1 PROTAC in vivo study workflow infographic (BOC Sciences).
Fig.1 PROTAC in vivo study workflow infographic (BOC Sciences).


