Project Background
A biotechnology company previously developed a water-soluble cyclic scaffold PROTAC molecule targeting intracellular proteins regulating the CDK4/6 signaling pathway in breast cancer. Later, the company aimed to apply this molecule for in vivo degradation studies in drug-resistant breast cancer, seeking to improve intracellular delivery efficiency and in vivo degradation performance. Due to technical challenges in PROTAC in vivo targeting and antibody conjugation stability, they engaged BOC Sciences for professional design optimization support.
Technical Challenges
Preliminary experiments showed that although the original PROTAC could degrade the target protein in vitro, it had limited cell penetration in vivo. Additionally, antibody conjugation suffered from stability issues and non-uniform DAR distribution.
BOC Sciences Solutions
- PROTAC Molecule Optimization: Introduced multiple water-soluble groups and employed ring expansion, contraction, and electronic steric strategies to fine-tune the scaffold, enhancing solubility, intracellular penetration, and stability while retaining E3 ligase recruitment activity.
- Linker Optimization: Designed and evaluated multiple controllable cleavable linker strategies, ensuring efficient intracellular release of the PROTAC payload while maintaining antibody structural integrity.
- Antibody Engineering: Screened multiple IgG1 antibodies suitable for site-specific conjugation, combined with cysteine site engineering to achieve controlled payload attachment and maintain antibody stability.
Project Outcomes
We designed and synthesized over 30 different PROTAC molecules and systematically evaluated each in combination with various antibodies and linkers. The optimized PAC featured a water-soluble PROTAC core and achieved over 80% target protein degradation in HER2/CDK4/6 drug-resistant breast cancer cells. Intracellular delivery efficiency was significantly improved, antibody structure remained stable, DAR distribution was uniform, and off-target effects were reduced.
Project Background
A biopharmaceutical company was developing a STAT3-targeting PROTAC payload to treat solid tumors with high EGFR expression. Although the core PROTAC molecule exhibited strong catalytic degradation in cell-free assays, it faced poor membrane permeability and rapid systemic clearance in animal models. To achieve tissue-specific delivery, the company sought to conjugate the STAT3 PROTAC with an anti-EGFR monoclonal antibody and engaged BOC Sciences to address complex three-component assembly and low conjugation yield challenges.
Technical Challenges
Key challenges include balancing payload hydrophobicity to prevent antibody aggregation, ensuring linker plasma stability, and maintaining antibody binding affinity throughout the conjugation process.
BOC Sciences Solutions
- PROTAC Payload Refinement: Redesigned the STAT3 PROTAC by introducing branched PEG chains and polar functional groups, reducing hydrophobicity while maintaining high affinity for the STAT3 SH2 domain and VHL E3 ligase.
- Dual-Stage Linker Engineering: Developed a Cathepsin B-responsive dipeptide linker with a self-cleaving spacer, ensuring the PACs were highly stable in plasma (pH 7.4) and released the payload rapidly and cleanly in acidic lysosomal environments.
- Enzymatic Site-Specific Conjugation: Utilized the MTGase enzymatic conjugation platform to precisely anchor the PROTAC payload onto specific glutamine residues of the antibody heavy chain, achieving DAR = 2.0 with high uniformity. SPR analysis confirmed preservation of antibody structure and binding kinetics.
Project Outcomes
BOC Sciences successfully delivered a lead PAC candidate with high uniformity and stability. The optimized conjugate achieved nanomolar STAT3 degradation (90%) in EGFR-high tumor cell lines. In xenograft models, PAC tumor concentrations were 5-fold higher than the unconjugated PROTAC, with markedly improved safety profiles, supporting the client’s progression into preclinical safety evaluation.

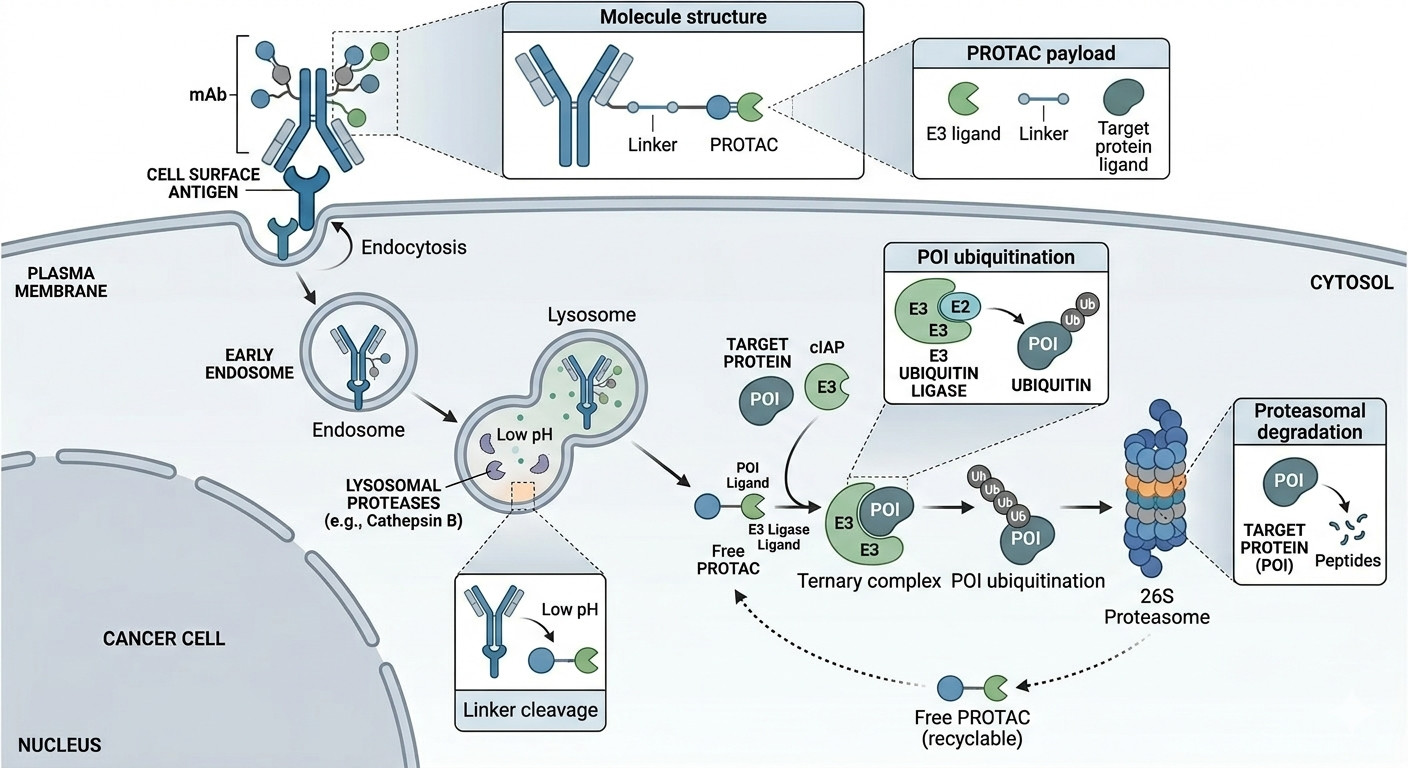 Fig.1 Mechanism showing PACs endocytosis and PROTAC-mediated protein ubiquitination (BOC Sciences).
Fig.1 Mechanism showing PACs endocytosis and PROTAC-mediated protein ubiquitination (BOC Sciences).

