The linker often determines whether an Aptamer-PROTAC can merely be conjugated or can truly function as intended. Its length, flexibility, hydrophilicity, attachment position, and cleavable or non-cleavable nature can all influence intracellular release after uptake, ternary complex formation, and overall degradation efficiency. In Aptamer-PROTAC systems, this issue is even more complex because the linker must also balance nucleic acid exposure with the productive conformation of the PROTAC moiety. In development practice, teams usually compare a matrix of short, medium, and long linkers, as well as flexible, semi-rigid, cleavable, and non-cleavable options, rather than relying on a single linker design.

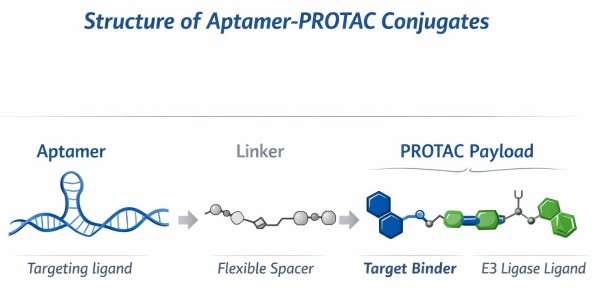 Fig.1 Aptamer-PROTAC conjugate structural composition schematic (BOC Sciences).
Fig.1 Aptamer-PROTAC conjugate structural composition schematic (BOC Sciences).


