Seasonal influenza caused by viruses has brought social and economic problems to the world. Fortunately, vaccines are one of the most effective means to prevent and control it. Among various vaccines, attenuated vaccine attracts much attention.
As the name suggests, the key to attenuated vaccines is "attenuated." Since viral replication requires key proteins encoded by the virus, the use of a host cell protein degradation system to regulate the stability of viral proteins may be an effective way to change the switch of the viral life cycle to achieve an attenuated virus. On July 4, a proteolysis-targeting chimeric virus vaccine (PROTAC vaccine) was reported by Chinese scientists. In this study, the wild-type virus was successfully attenuated into a safe vaccine by degradation of the viral protein by PROTAC.
PROTAC viruses bind the proteasome targeting domain (PTD) to influenza virus proteins. Structurally, the PTD contains proteasome targeting sequence (ALAPYIP) and tobacco etch virus cleavage site (TEVcs) linker group (ENLYFQG). Previous studies have fully demonstrated that ALAPYIP is recognized by VHL proteins to initiate polyubiquitin and proteasomal degradation of downstream target proteins, thus PTD can specifically induce degradation of target viral proteins. The TEVcs linker can be selectively cleaved by tobacco plaque virus protease (TEVp) to separate viral protein from PTD and avoid the degradation of viral protein by the PROTAC vaccine.
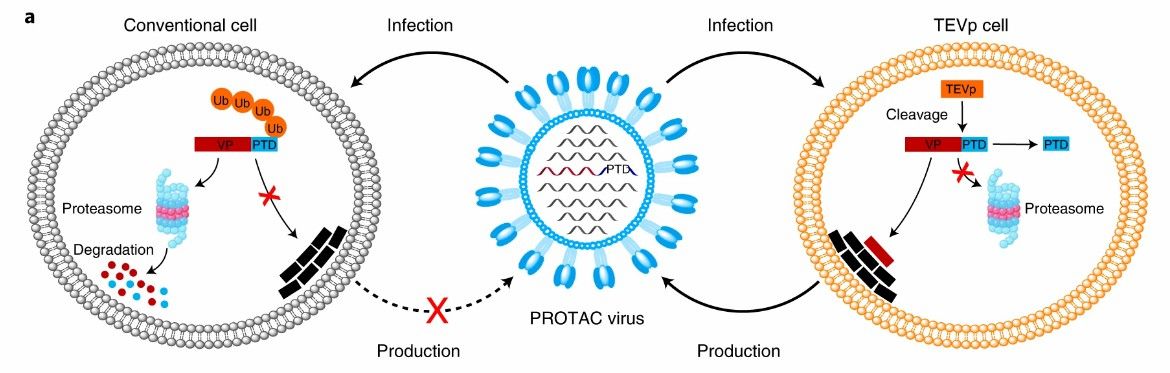 Fig. 1 Schematic illustration of the generation of PROTAC viruses (Si, 2022)
Fig. 1 Schematic illustration of the generation of PROTAC viruses (Si, 2022)
M1 is a viral protein. The PROTAC influenza virus vaccine M1-PTD constructed based on M1 breaks the linker group in the cell line statically expressing TEVp, and M1 is free from PTD without degradation, thus achieving high efficiency of virus replication in the process of vaccine preparation. While in normal cells, the M1-PTD viral protein will be degraded, thus achieving viral attenuation.
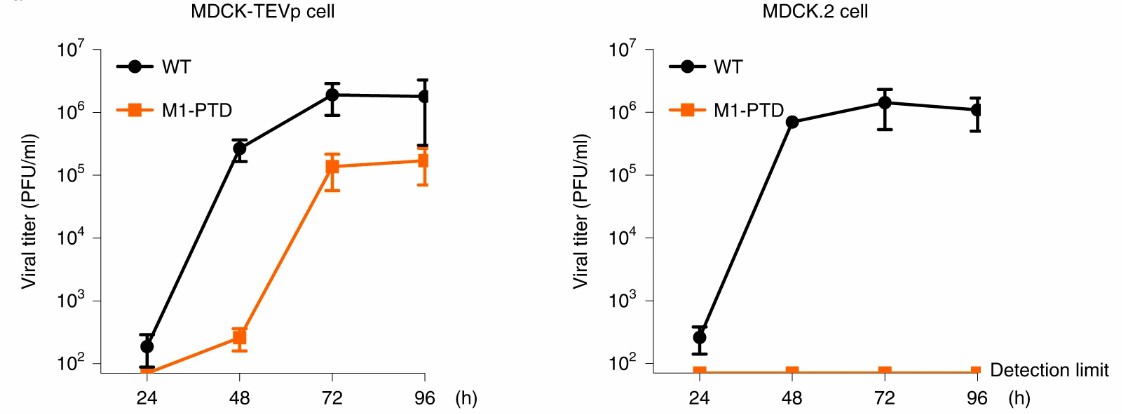 Fig.2 M1-PTD replicates efficiently only in TEVp stably expressing cell lines (Si, 2022)
Fig.2 M1-PTD replicates efficiently only in TEVp stably expressing cell lines (Si, 2022)
In the mechanism study, the proteasome inhibitor MG-132 could improve the efficiency of viral replication and restore the level of M1 protein. Inhibition of VHL activity by VH298 also reduced M1 protein degradation. Therefore, the above studies indicated that the degradation mechanism of the PROTAC virus was VHL-mediated proteasome degradation.
VHL protein is ubiquitously expressed in most normal tissues and cells, which provides an important basis for the safety of the PROTAC vaccine. Virus plaque is a localized lesion formed by a virus on mature monolayers of cells. M1-PTD could only form plaque in cell lines with stable expression of TEVp, but this phenomenon was not observed in normal cells. The safety of M1-PTD was further evaluated in animal models. Mice were inoculated with 104 PFU (plaque forming unit) of wild-type virus and 105 PFU (M1-PTD) virus, respectively. The survival rate and body weight of mice were monitored. The data showed that the M1-PTD virus did not cause death in mice, and there was no significant change in body weight. Thus, M1-PTD successfully attenuated wild-type virus into a safe influenza vaccine.
M1-PTD can induce a wide range of immune responses in vivo. The results of the immunological evaluation showed that M1-PTD activated humoral, mucosal, and cellular immune responses at the same time, and could provide powerful protection against homologous and heterologous viruses.
Previously developed PROTAC tends to exert cytotoxic effects by degrading disease-causing key proteins. A recent study published in the journal Cell explored the possibility of using PROTAC to degrade target proteins in bacteria, which is expected to be a new direction for the development of antimicrobial agents. This study further broadens the application scenario of PROTAC and provides a new idea for the development of a virus vaccine.
As a proof of concept, M1-PTD demonstrates the feasibility of PROTAC as a viral vaccine. However, the PROTAC vaccine still has a long way to go, as the experiments were carried out at the cellular and animal levels.
References
- Longlong Si. Generation of a live attenuated influenza A vaccine by proteolysis targeting. Nature Biotechnology. 2022.

 Fig. 1 Schematic illustration of the generation of PROTAC viruses (Si, 2022)
Fig. 1 Schematic illustration of the generation of PROTAC viruses (Si, 2022) Fig.2 M1-PTD replicates efficiently only in TEVp stably expressing cell lines (Si, 2022)
Fig.2 M1-PTD replicates efficiently only in TEVp stably expressing cell lines (Si, 2022)