Since RTKs play crucial roles in cancer development, targeting oncogenic driver mutations of RTKs has revolutionized the treatment of cancer patients. RTK-targeting PROTAC is one such emerging therapy. As a leading CRO in targeted protein degradation, BOC Sciences provides one-stop RTK PROTACs design services to meet our clients' new drug discovery goals.
Introduction
Receptor tyrosine kinases (RTKs) play key roles in signaling pathways that regulate intercellular communication and control essential cellular processes. Thus, dysregulation of RTK signaling contributes to a wide variety of human diseases, particularly cancer. RTKs are a subclass of tyrosine kinases and there are approximately 58 known RTKs in humans, including EGFR, HER2 and MET, all of which have similar protein structures. Under normal physiological conditions, RTKs are activated in a ligand-mediated manner to propagate cellular signals. Oncogenic mutations in RTKs disrupt this normal biological process, leading to dysregulated cell growth, abnormal cell signaling, and altered tumor cell metabolism. Overexpression of RTKs has been found in a variety of human cancers, such as EGFR in glioblastoma, lung, esophageal thyroid cancers; HER2/ErbB2 in lung, bladder, breast and gastric cancers; and MET in lung and gastric cancers. Because of the critical role of RTKs, many small molecule inhibitors or monoclonal antibodies have been developed to treat cancers and other diseases caused by mutations in RTKs. Despite these advances, acquired resistance to targeted therapies will inevitably occur. The proteolysis targeting chimera (PROTAC) technology, as a next-generation treatment, has the potential to overcome these concerns.
About RTK PROTACs
PROTAC is a heterobifunctional small molecule, consisting of two ligands, one for the target protein and the other for an E3 ubiquitin ligase. RTK PROTACs bind to the target (in this case RTK) and the E3 ubiquitin ligase simultaneously, which drives the polyubiquitination and the subsequent degradation of the target protein. Unlike the conventional small-molecule inhibitors, PROTACs eliminate both catalytic and noncatalytic (scaffolding) functions of the targeted enzyme. The advantages of degradation induced by RTK PROTACs over inhibition include more potent inhibition of cell proliferation, a more durable and sustained downstream signalling response, so RTK PROTACs address the kinome re-wiring challenge seen with many RTK inhibitors. Indeed, the elimination of RTK would prevent the persistence of inactive kinases as scaffolding nodes for oncogenic signaling, and this more sustained loss of function could be accomplished using PROTACs, a technique for post-translational protein degradation.
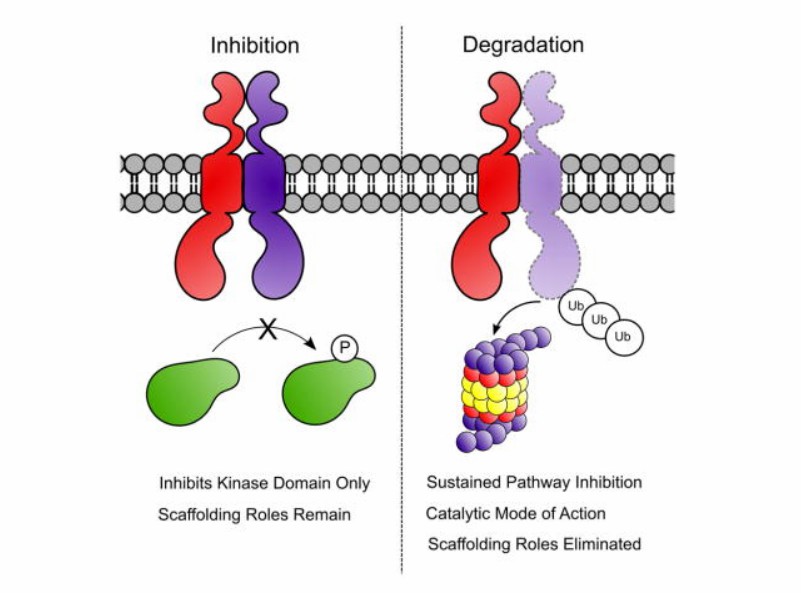 Fig. 1 Comparison of degradation and inhibition (Burslem, 2018)
Fig. 1 Comparison of degradation and inhibition (Burslem, 2018)
Application
Unlike kinase activity inhibition, RTKs degradation results in complete, lasting downstream signal inactivation. PROTACs degraded several RTKs, including c-Met, HER2, EGFR, and multiple mutants of EGFR. PROTACs have many advantages such as improved physicochemical properties, reduced toxicity, and facile modular design. EGFR, as a kind of RTK, is critically associated with the regulation of cell apoptosis, proliferation, metabolism, and survival. Several PROTACs have been reported that conjugating an EGFR-binding ligand (commonly EGFR inhibitors, e. g. Gefitinib) to an E3 ligase binding ligand for EGFR degradation. EGFRL858R/T790M serves as an ideal and relatively safe target for PROTACs because it is selectively expressed in tumor cells with acquired drug resistance, but it can be found in normal cells.
Our Services
- Selection of RTK ligands
- Design and synthesis of RTK PROTACs
- Testing of RTK PROTACs cell membrane permeation
- Evaluation of the degradation level of RTK by PROTACs
Related Products
Our Advantages
- Custom RTK-targeting PROTAC development
- Comprehensive one-stop PROTAC platform
- Data analysis, detailed report with results and discussion
- Short turn-around time and competitive price
- Highly reliable and reproducible result
References
- Burslem, G. M., et al., The Advantages of Targeted Protein Degradation over Inhibition: a RTK Case Study, Cell Chem Biol., 2018, 25(1): 67-77.e3.
- Du, Z., and Lovly, C. M., Mechanisms of receptor tyrosine kinase activation in cancer, Molecular Cancer, 2018, 17, 58.

 Fig. 1 Comparison of degradation and inhibition (Burslem, 2018)
Fig. 1 Comparison of degradation and inhibition (Burslem, 2018)